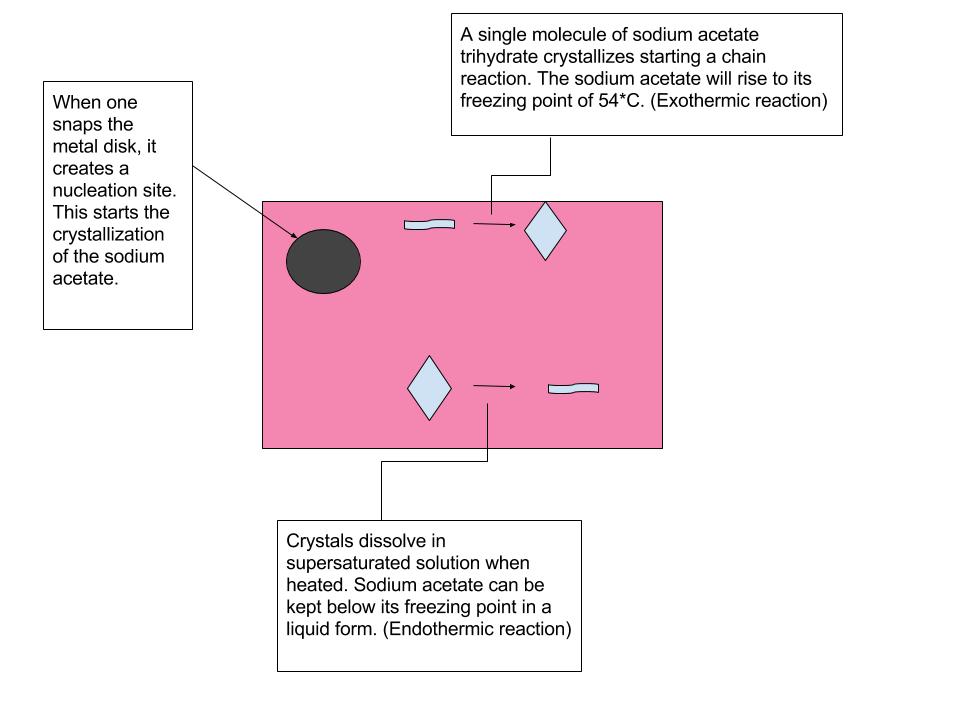
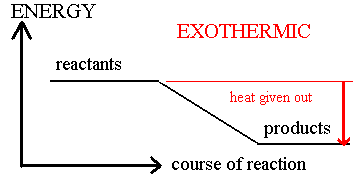
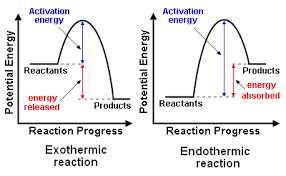
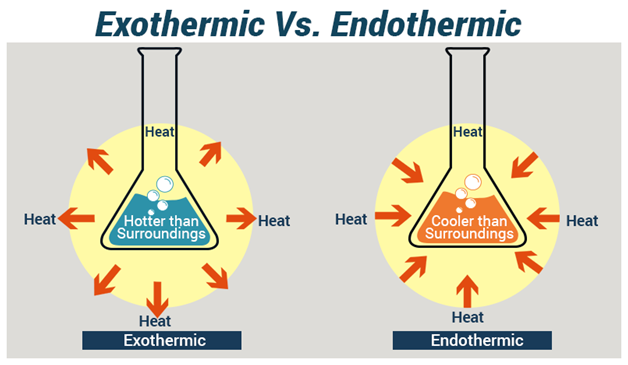
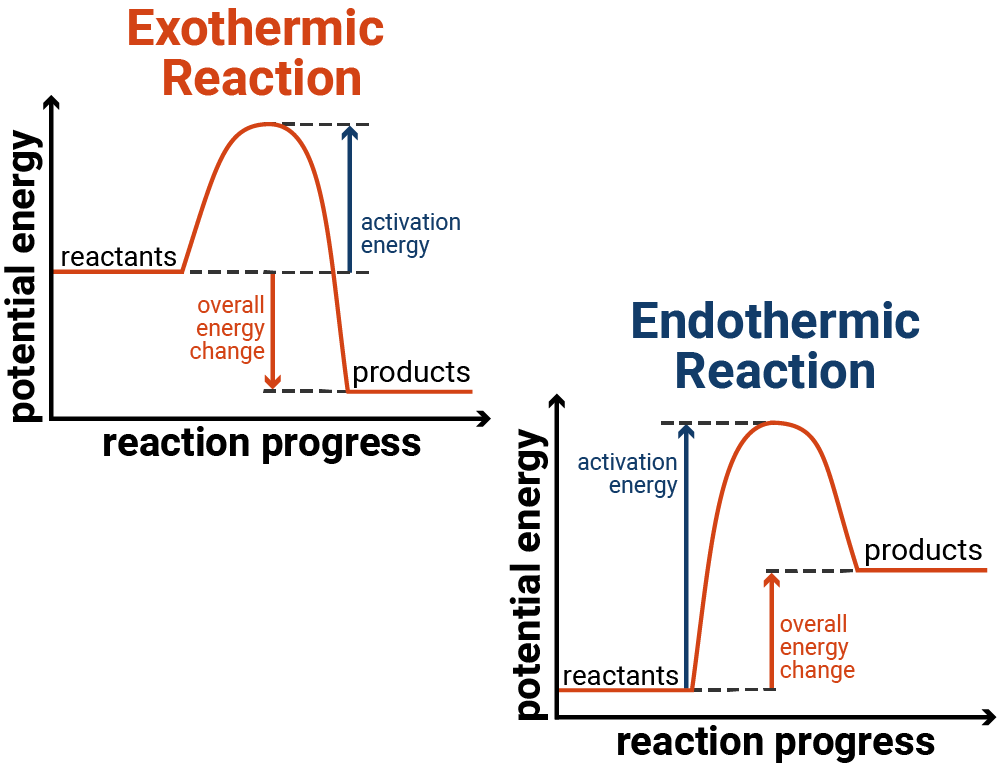
SOLVED: Which statement explains a difference between endothermic and exothermic reactions? A. Endothermic reactions release energy, while exothermic reactions absorb energy. B. Endothermic reactions feel cool to the touch, while exothermic reactions